Posted on Wednesday, March 04 2015 @ 12:47 CET by Thomas De Maesschalck
Could glass-coated sulfur particles become the next big thing in the battery industry? Research by the Bourns College of Engineering at the University of California, Riverside
suggests glass-coated sulfur particles may be the
breakthrough required to drive market adoption of lithium-sulfur batteries. If this technology succeeds, we may get batteries that offer ten times the energy density of current lithium-ion batteries.
A fresh, new lithium-sulfur battery can have energy density ten times that of a conventional lithium-ion battery. Imagine a large Android phone with 30,000mAh of juice instead of just 3,000mAh. How fantastic would that be? With current lithium-sulfur technology, that starts dropping off rather quickly as lithium and sulfur reaction products start clogging the works. These products, called lithium polysulfides, dissolve in the electrolyte solution and become stuck at the electrodes. This causes an overall decrease in capacity, and there’s no way to reverse it.
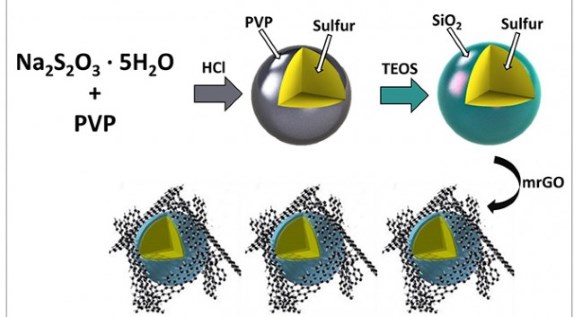
Sulfur particlesThe UC Riverside team found they could prevent this “polysulfide shuttling” by using nano-scale sulfur beads in the battery’s cathode and coating them with SiO2, which you might know as glass. The thickness of this silica sheath is measured in tens of nanometers. It can’t be too thick or it would interfere with the battery’s function. However, it also can’t be too thin, or the glass layer could rupture and allow the formation of lithium polysulfides that damage the structure.